SmartSyringe
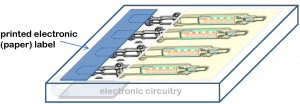
Syringes can be modified to enable electronic therapy compliance monitoring. An adhesive label is made to suit the dimensions of the existing syringe packaging. When a syringe is removed from the package an electronic track on the label is broken and this is detected by a small microprocessor chip that stores the time and date at which the syringe was taken. The data stored in this microprocessor can be read out using NFC (Near Field Communication) readers or even smart phones with the NFC feature and one of our apps.
